Overview of Hypercalcemia
Hypercalcemia refers to the presence of elevated calcium level above normal. The normal level of total serum calcium varies from 8.5 to 10.5 mg/dL or for ionized calcium 4.2 to 4.8 mg/dL.
When extracellular calcium is increased, the duration of the action potential is shortened. Shortening of the action potential duration results in shortening of the QT interval.
The ECG findings of hypercalcemia include (Figs. 1 and 2):
Shortening of the QT interval. This is due to shortening of phase 2 of the action potential corresponding to the ST segment in the ECG.
Elevation of the ST segment especially in the precordial leads. This may be mistaken for acute ischemic injury.
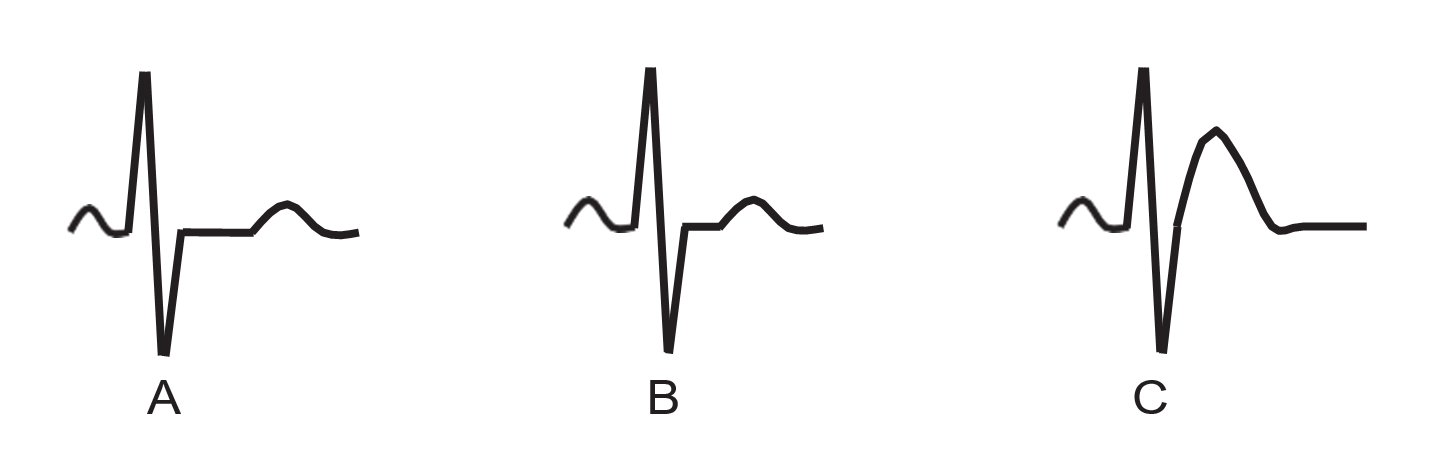

ECG Findings of Hypercalcemia
- Short QT interval from shortening of the ST segment
- Flattened and widened T wave with ST elevation
- Prolonged P-R interval
- Widened QRS complex
- Increased QRS voltage
- Notching of the terminal portion of the QRS complex from a prominent J wave
- AV block progressing to complete heart block and cardiac arrest when serum calcium >15 to 20 mg/dL.
Mechanism
- Increasing levels of serum calcium may cause changes in the ECG. Unlike hyperkalemia, in which the ECG changes are more dramatic, the ECG abnormalities associated with hypercalcemia are less specific and should not be used as the basis for making the diagnosis of hypercalcemia.
- Hypercalcemia shortens the duration of the action potential of the myocyte, resulting in a shortened QT interval. This usually occurs when the serum calcium is >13 mg/dL. Because the duration of ventricular systole is shortened, ventricular refractoriness is shortened, rendering the
- Figure 2: The ST and T Wave Configuration in Hypercalcemia. Total calcium level is 16.0 mg/dL. Note the short ST segment and ST elevation in V3 to V6. Prominent J wave or Osborn wave may also occur when there is hypercalcemia.
- patient more prone to arrhythmias. It also renders patients more susceptible to the toxic effects of digitalis.
- Hypercalcemia initially increases inotropicity and chronotropy by causing increased calcium influx and decreases calcium egress in the myocyte. However, as serum calcium further increases to levels >15 to –20 mg/dL, myocardial contractility becomes depressed.
- Very high levels of calcium, >15 to 20 mg/dL, may result in arrhythmias most commonly bradycardia and complete heart block.
Clinical Significance
- Calcium is the most common mineral in the body; 99% of the total amount of calcium is stored in bones. The remaining 1% is distributed in the sera, 50% of which is bound to albumin and the rest available as ionized calcium. Total serum calcium is affected by the level of serum albumin. When serum albumin is low, total calcium is low. Inversely, when serum albumin is high, the total calcium level is high. The level of ionized calcium, however, is not affected by the level of serum albumin and is more important in causing signs and symptoms of calcium excess or deficiency.
- Pseudohypercalcemia can occur when there is profound dehydration, causing increased binding of calcium by albumin. This will result in increased total serum calcium but the level of ionized calcium remains normal. This can also occur in some patients with multiple myeloma.
- The normal level of total serum calcium is 8.5 to 10.5 mg/dL and of ionized calcium 4.2 to 4.8 mg/dL. Hypercalcemia indicates the presence of high levels of serum calcium above the normal range. When real hypercalcemia is suspected, the level of ionized calcium should be checked. Ionized calcium is collected anaerobically, adjusted to a normal pH of 7.4, and is often reported as normalized calcium.
- The level of ionized calcium is actively regulated by the endocrine system. When there is hypocalcemia, enhanced secretion of parathyroid hormone (PTH) increases osteoclastic activity and bone resorption, thus increasing the level of calcium in the blood. PTH also promotes absorption of calcium in the GI tract by activating Vitamin D and decreases excretion of calcium in the kidneys by promoting tubular reabsorption. Inversely, when there is increased level of calcium, PTH secretion is inhibited and calcitonin is released, which will lower serum calcium by reducing osteoclastic activity and increasing the deposition of calcium in the bones and at the same time increase excretion of calcium by the kidneys.
- Ionized calcium is affected by pH, whereas total calcium is not. When there is metabolic or respiratory alkalosis, H+ is shifted from plasma proteins to serum to buffer the increased bicarbonates. More ionized calcium will become proteinbound to neutralize the more negatively charged plasma protein, thus decreasing the level of ionized calcium. The reverse happens when there is acidosis: ionized calcium increases in the serum.
- The two most common causes of hypercalcemia accounting for more than 90% of cases are hyperparathyroidism and malignancy.
- Hyperparathyroidism may be primary from an autonomously hyperfunctioning parathyroid gland (primary hyperparathyroidism) or secondary from chronic renal disease (secondary hyperparathyroidism).
- Hypercalcemia of malignancy is due to increased osteoclastic activity, resulting in increased bone resorption. This may be due to increased hormonelike substances in the blood or direct invasion of tumor cells into the bone.
- Humoral hypercalcemia: from increase in PTH-like substances in the blood, resulting in increase osteoclastic activity and bone resorption. This type of hypercalcemia is seen in squamous cell cancer of the lungs, head and neck, and often renal and ovarian cancer.
- Bone metastasis: direct bone metastasis may also result in increased bone resorption most commonly the result of breast cancer or multiple myeloma.
- Other causes of hypercalcemia include use of drugs such as thiazide diuretics, lithium, and vitamins A and D or sarcoidosis and other granulomatous diseases.
- There are usually no physical findings associated with the hypercalcemia itself. Symptoms of hypercalcemia usually do not occur until the serum calcium reaches 12 mg/dL or higher. Hypertension is common in patients with hypercalcemia and is a common manifestation in patients with primary hyperparathyroidism. At serum levels of 12 to 15 mg/dL, weakness, apathy, fatigue, depression, and confusion may occur. GI symptoms of constipation and dysphagia are common.
- A higher incidence of dyspepsia and peptic ulcer disease may occur because of a calcium-mediated increase in gastrin secretion. As hypercalcemia becomes more severe, dehydration may occur because hypercalcemia decreases renal concentrating capacity, resulting in polyuria and polydipsia. Finally, neurologic symptoms characterized by hallucinations, disorientation, and coma may develop. Although symptoms of hypercalcemia are usually neuromuscular, cardiac manifestations may occur, including AV block and cardiac arrest.
Treatment of Hypercalcemia
- Treatment is directed toward the underlying cause of the hypercalcemia. Therapy for hypercalcemia is essential when the calcium level is >12 mg/dL, especially when the patient is symptomatic. Therapy is mandatory at levels >15 mg/dL, regardless of symptoms.
- Excessive increase of calcium in the blood causes polyuria and GI symptoms, especially in patients with malignancy, resulting in dehydration. This enhances reabsorption of calcium in the kidneys, thus further worsening hypercalcemia. Patients with hypercalcemia are therefore volumecontracted; proper hydration with restoration of extracellular volume promotes calcium excretion.
- Saline diuresis: In symptomatic patients with severe hypercalcemia >15 mg/dL who have reasonably preserved cardiovascular and renal function and are dehydrated, the 2005 AHA guidelines for cardiopulmonary resuscitation and emergency cardiovascular care recommends intravenous infusion of 300 to 500 mL/hour of 0.9% saline until any fluid deficit is corrected or until patient starts to diurese adequately. After the patient is properly hydrated, IV hydration is continued at 100 to 200 mL/hour to maintain adequate diuresis and promote calcium excretion. At least 3 to 4 L is usually needed in the first 24 hours. Other electrolytes, especially potassium and magnesium, should be monitored carefully.
- Loop diuretics: Loop diuretics (e.g., furosemide [20–40 mg 2 to 4 times daily] or bumetanide [1 to 2 mg twice daily]), may be used in patients with heart failure, although their use in the treatment of the hypercalcemia itself is controversial and should be used only after appropriate volume repletion with normal saline. Thiazide diuretics should not be substituted for loop diuretics because they prevent calcium excretion.
- Calcitonin: Calcitonin lowers serum calcium by inhibiting bone resorption and promoting urinary calcium excretion.
- Bisphosphonates: Biphosphonic acid lowers serum calcium by inhibiting osteoclastic bone resorption. The following bisphosphonates are commonly used in the treatment of hypercalcemia associated with malignancy.
- Pamidronate: Pamidronate is given as an intravenous infusion. It can be combined with calcitonin to provide a longer effect. There is risk of renal toxicity if given rapidly or in high doses.
- Zoledronic acid: Zoledronic acid is preferred as it is more potent than pamidronate. It can be infused over a shorter period. The drug can also cause renal damage if the infusion is given rapidly or in high doses. Renal function should be reassessed if a second infusion is necessary. Patients receiving the infusion should be properly hydrated.
- Steroids: Glucocorticoids reduce calcium level by several mechanisms. They inhibit intestinal absorption, increase uri nary excretion of calcium, and have cytolytic effect to some tumor cells, especially multiple myeloma and other malig- nancies. They also inhibit calcitriol production by mononu- clear cells in lungs and lymph nodes; thus, they are effective in hypercalcemia associated with granulomatous diseases and occasionally with lymphoma.
- Phosphates: Phosphates are given orally to prevent calcium absorption. It combines with calcium to form complexes that limits its absorption. It also increases calcium deposition in bones.
- Hemodialysis: Hemodialysis should be considered when there is need to promptly decrease the level of serum calcium in patients with heart failure or renal failure who cannot tolerate saline infusion. The dialysis fluid should be altered because the conventional dialysis solution may have a composition that may not be ideally suited for rapid correction of the electrolyte abnormality.
Prognosis
- Prognosis depends on the underlying condition. Hypercal- cemia is commonly associated with malignancy; thus, the in- tensity of therapy should be individualized and should con- sider the overall clinical picture.
